 
Noble gases on the far right do not have any charges. 
(a) The covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. Starting with a charge of positive one for group 1, positive 2 for group 2, and negative 4, 3, 2, and 1 for groups 14, 15, 16, and 17 respectively. Atomic radii are often measured in angstroms (), a non-SI unit: 1 1 × 1010 m 100 pm. This periodic table shows the charges of ions corresponding with the group number. In the molecule CoN, Cobalt has a +3 charge because Nitrogen forms a -3 charge, and to make the molecule neutral, Co has to be +3.\): Some elements exhibit a regular pattern of ionic charge when they form ions. For example, in CoBr2, we see that Cobalt is +2 because Bromine is a +1 ion, and two Bromines create a charge of +2. Atoms in the lower left part of the periodic table easily become cations. D-orbitals have a variety of oxidation states, whereas the s-orbitals in the alkaline earth metals cause these metals to have set oxidation states. Examples include the alkalis, such as sodium and potassium. 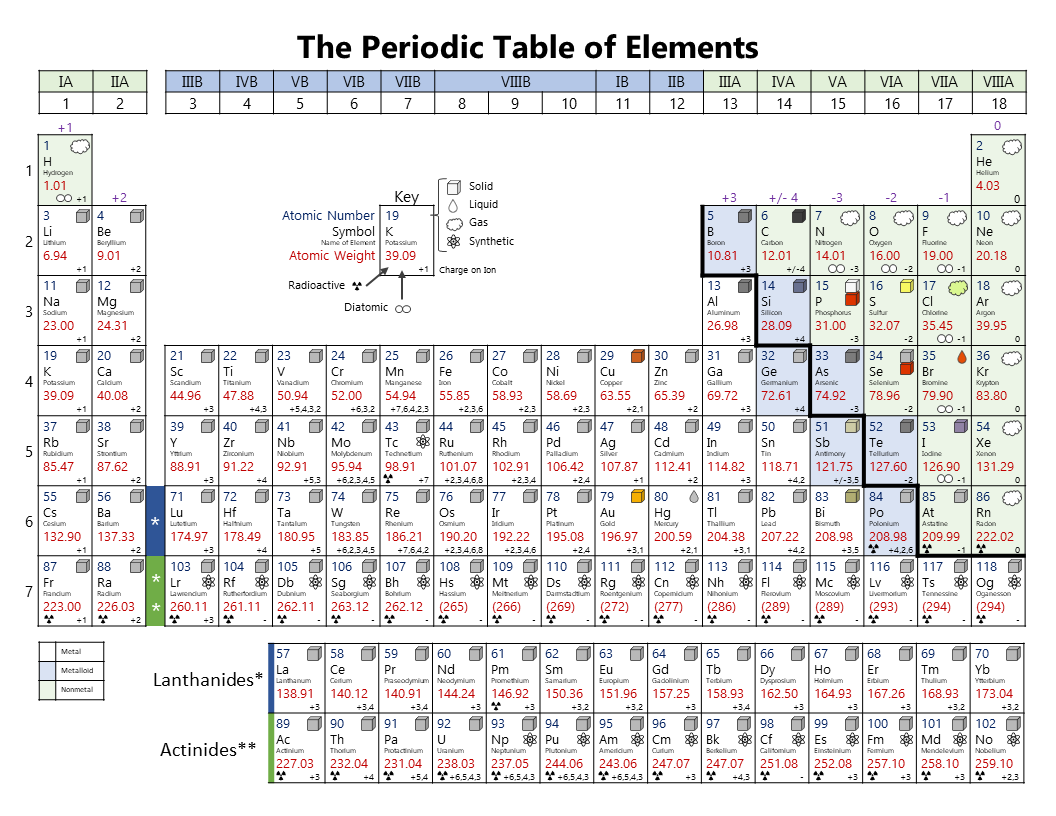
This is because transition metals have 5 d-orbitals. There is a quick way to calculate the charge on an ion: the number of charges on an ion formed by a metal is equal to the metals group number. You can use the periodic table to predict whether an atom will form an anion or a cation, and you can often predict the charge of the resulting ion. Why does this happen? Because it is a lot easier for transition metals to lose electrons compared to the alkaline earth metals (groups one and two). Though we can see that transition metals form positive ions, and some transition metals have set charges, most transition metals are extremely variable, and depending on what element they bond with, they can become one of many different ions. As discussed in class, the elements in the first column of the periodic table (excluding hydrogen) always lose one electron to get a +1 charge. What is the charge on its ions, and is the charge. 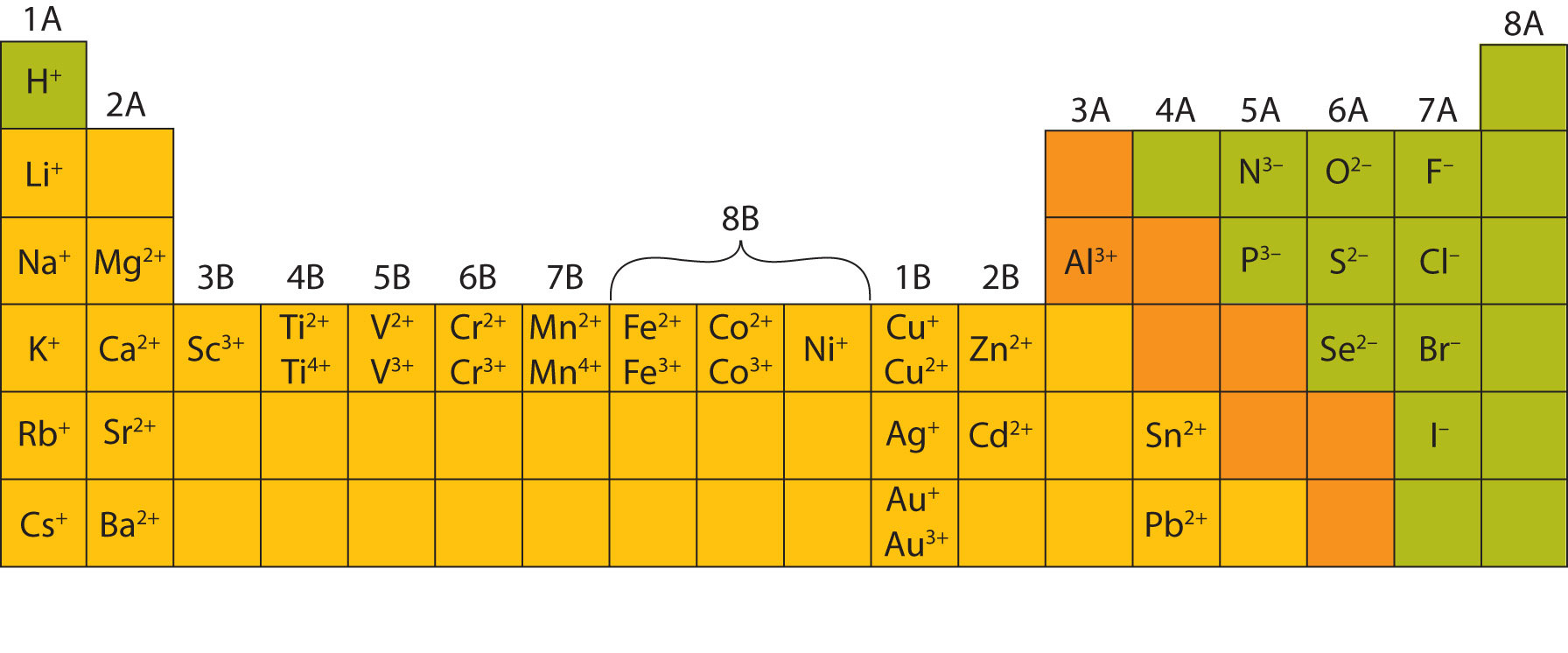
Sulfur is in group 6 of the periodic table. Group 17 elements, the halogens, form -1 ions, and group 16 elements form -2 ions because they need to give away one and two electrons to have a full outermost electron orbit respectively. Element: O: Ion charge: 2-Ion symbol: O 2-Group: 7: Element: Cl: Ion charge-Ion symbol: Cl-Example. Group 18 elements, the noble gases, do not form ions. This implies that the atoms of elements in. This is because groups one and two need one and two electrons to complete their outermost electron orbit respectively. The group number of an element in the periodic table indicates the number of the electrons in its outermost orbit. For example, copper usually has a +1 or +2 valence, while iron typically has a +2 or +3 oxidation state. For example, iron(II) has a 2+ charge iron(III) a 3+ charge. Metal ions may have other charges or oxidation states. Roman numeral notation indicates charge of ion when element commonly forms more than one ion. Family, or group, one elements form +1 ions while group two elements from +2 ions. Group I ( alkali metals) carry a +1 charge, Group II (alkaline earths) carry a +2, Group VII (halogens) carry -1, and Group VIII ( noble gases) carry a 0 charge. When we look at the periodic table, we can observe the placement of many elements to find out the charge of its ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
